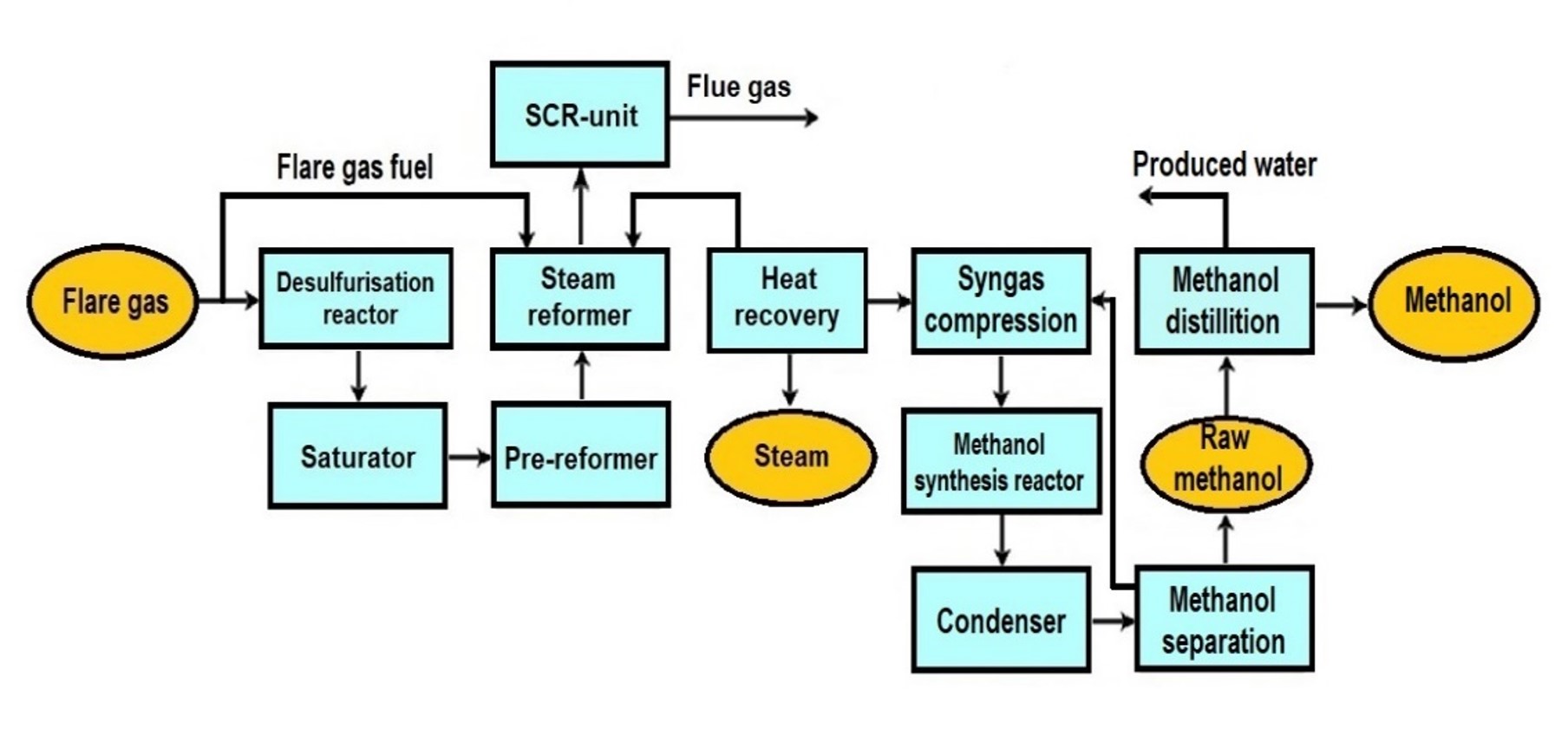
General Overview
Worldwide, over 90 methanol plants have a combined production capacity of about 110 million metric tons (almost 36.6 billion gallons) .
The first methanol production from synthesis gas (syngas) was established in the German Leuna Works, a large chemical factory near the city of Merseburg, by the BASF physical chemist Matthias Pier (1882 – 1965), a pupil of the German Nobel Prize Laureate Walther Nernst, already in 1923.
The BASF process worked at a pressure at above 300 bar and a temperature of 300 up to 400 oC and was using, remarkably for this time, solid catalyst after the recipe of Alwin Mittasch (1896 – 1953), which was basically a mixture of Zn(II)oxide and Cr(III)oxide.
Syngas is composed of hydrogen (H2), carbon monoxide (CO) and carbon dioxide (CO2), whereas the ration H2/CO is important in view of the process efficiency using a certain catalyst material.
Step 1: feed purification
After the natural gas (feed gas) is compressed (see figure above) the desulfurisation occurs (warm clean-up) commonly in a fixed-bed reactor with zink(II) oxide by simply following the chemical reaction:
ZnO(s) + H2S(g) à ZnS(s) + H2O(g) (1)
Consequently, the catalyst also can be regenerated, as follows:
ZnS(s) + O2(g) à ZnO(s) + SO2(g) (2)
Another process, commonly used in refineries and applied for the removal of sulfur is hydrodesulfurisation (HDS). Most methanol processes applied today use the steps outlined below.
Downstream of the desulfurisation reactor, the desulfurised gas will be cooled down in order to enter the saturator where it is treated with hot water in a kind of packed bed column. Owing to that treatment, the saturated gas already is enriched with a certain amount of the steam which later is required for the reforming.
Step 2: Reforming / syngas generation
Syngas is produced from natural gas / gas through a reforming process, which is commonly Steam Methane Reforming (SMR), where additional steam will be added in order to achieve the necessary “steam to carbon ratio” for the reforming process step.
In the SMR process step, methane reacts with steam under 3-25 bar pressure (1 bar= 14.5 psi) in the presence of a catalyst to produce hydrogen and carbon monoxide:
CH4(g) + H2O(g) ⇌ CO(g) + 3 H2(g) (3)
Commonly a comparatively small amount of carbon dioxide is also generated.
Other reforming processes are Partial Oxidation (POX) and Autothermal Reforming (ATR).
Step 3: Methanol synthesis
The synthesis gas is fed to the methanol synthesis converter at about 130° C. The converter is of different design (typically in cascades) depending on the particular technology applied. The compressed gas is preheated to reaction temperatures inside the tubes as it flows through the hot catalyst bed.
The hot reacted gas leaves the converter and provides heat to the saturator water circuit and the loop interchanger before finally being cooled. Crude methanol is separated from the uncondensed gases and the gases are recirculated back to the converter via the circulator.